The Immediate Fate of Abortion Access Lies in the Hands of One Texas Judge
The fight over reproductive rights has dominated legal debate and public discourse as of late. But unlike this time last year, when the United States Supreme Court was the battleground for a high-stakes dispute over abortion, the fate of these rights now heavily rests in the hands of a single judge in Amarillo, Texas.
Before U.S. District Judge Matthew Kacsmaryk sits a case concerning mifepristone, an abortion medication used by an increasing number of patients to terminate pregnancy. The lawsuit, brought by anti-abortion group Alliance for Hippocratic Medicine, asks the Donald Trump-appointed judge to order the Food and Drug Administration to repeal its approval of the drug, effectively pulling it from the market and altering the state of medication abortion nationwide.
Medication abortion, an alternative to the traditional surgical procedure, accounted for more than half of abortions performed in the U.S. in 2020, and the method has taken on even greater significance in a post-Roe world as a growing number of states restrict or ban abortion. Mifepristone, the drug at the center of the Texas case, is used in 98% of these abortions—alongside a second medication, misoprostol—to terminate pregnancies at up to 10 weeks.
The lawsuit alleges, however, that the medication’s approval by the FDA more than 20 years ago was not properly evaluated and should not have made mifepristone available via telehealth during the COVID-19 pandemic. If Kacsmaryk rules in favor of the case, granting the request for a preliminary injunction to remove mifepristone from the market, it could result in a nationwide ban on the widely utilized abortion pill—even in safe-haven states, like California and New York.
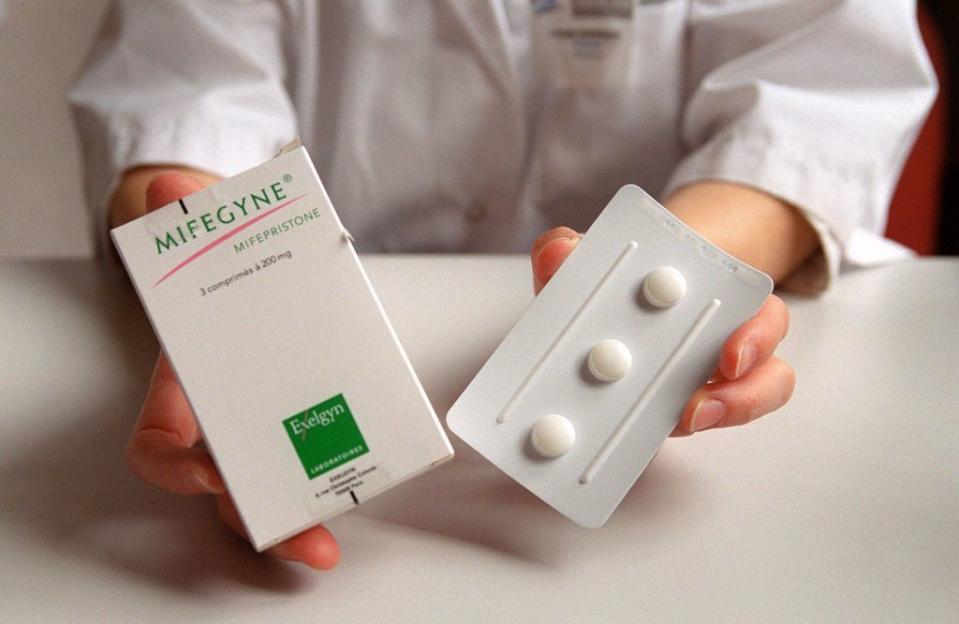
“It was an arduous process to get [mifepristone] approved,” Melissa Grant, the cofounder and CEO of abortion care provider Carafem, tells BAZAAR.com. The drug, which was developed in France in 1980 and rolled out across Europe in the subsequent decade, became the subject of three clinical trials in the U.S., one of which she participated in early in her career. “In 1996, more than 15 years after mifepristone came out, the FDA finally decided to go ahead and approve it, and it still sat for another four years before it was finally approved,” Grant says. “So, it’s white noise and political maneuvering to say that this drug was approved in some way that was not thoughtful. It’s a politically motivated decision instigated by extremists.”
The Texas case comes as just a small piece in a much bigger attack on abortion, one that abortion providers and activists have been fending off for years. This only escalated during the pandemic, when suddenly, patients were confined to their homes and health care was mostly limited to telemedicine. Even so, some in the field saw these difficulties as ample opportunity to expand abortion access.
“We were seeing digital health companies focusing on stigmatized issues in men’s health take off and I thought, ‘This could be a model for safe, discreet, affordable abortion access,’” recalls Kiki Freedman, cofounder of Hey Jane, a virtual clinic that offers telemedicine abortion care. Since 2021, the provider has helped 25,000 patients receive private, safe, supportive, accessible and convenient care—largely with the help of mailed medication abortion.

The pandemic initially allowed for this expansion of medication abortion access, when the in-person dispensing requirement for abortion pills was lifted in the summer of 2020, but a challenge by the Trump administration in January 2021 soon reinstated the in-person requirement. A few months later, the FDA decided not to enforce the requirement for the duration of the pandemic, and in December 2021, the suspension was made permanent. But many abortion providers saw the writing on the wall and knew that this wouldn’t be the end of the telehealth medication abortion discussion.
The Dobbs decision last June, which immediately made abortion illegal or extremely difficult to access in a number of states, has only heightened the importance of medication abortion. Patients living in states where abortion is restricted have found ways to obtain medication abortion from across state lines, whether through legal means or through alternative routes without clinician involvement. “In states that unjustly restrict access to abortion care through mainstream medical clinics, we know that abortion pills obtained through alternate sources have played a huge role in helping people access safe abortion,” says Elisa Wells, the cofounder and co-director of Plan C, a non-profit organization and campaign for access to medical abortion. “We estimate that just since the Dobbs decision, 30,000-40,000 people or more have obtained abortion pills through these alternate sources and safely self-managed their abortions.”
If Judge Kacsmaryk does rule in favor of the Texas lawsuit, as he is expected to any day now, mifepristone could be pulled from the market, leaving many patients without a safe or accessible path to terminate pregnancy, even in states where surgical abortion remains available. “I couldn’t afford a surgical abortion, and I didn’t want it to show up on my insurance since I didn’t tell my parents at the time,” recalls Hannah Partin, who at 22 years old underwent a dual-medication abortion in New York City. “I think not having medication abortion as an option would have really impacted my thinking around having an abortion, even if it wouldn’t have changed the outcome.”

Many patients may soon find themselves in similar positions if the Texas ruling on mifepristone forces abortion providers to stop offering the foremost medication abortion method.
It is unclear what long-term impact the decision will have, and some in the health care space remain hopeful that FDA law will take precedent over Kacsmaryk’s decision. But even with the future of mifepristone unknown, many abortion providers have begun preparing for the worst and are creating alternative treatment plans to offer patients.
For Carafem, which began offering telemedicine abortion care in 2017, it was obvious back in 2020 that mifepristone was not always a perfect solution. “We’ve always tried to predict possibilities around decreasing access, and we found during the Trump administration that there was threat to the ongoing ability to mail mifepristone,” Grant says. “So, in 2020, we implemented a second option for medication abortion, in addition to the gold standard of mifepristone and misoprostol together, and that was to provide medication abortion through misoprostol alone.” In countries around the world where mifepristone is not available, misoprostol is the most commonly used medication and can successfully end early pregnancy up to 95% of the time. Carafem has continued to offer misoprostol alone as a secondary option and is prepared to shift to misoprostol exclusively if forced; it’s just one of several routes that abortion providers could take in response to the imminent Texas decision.
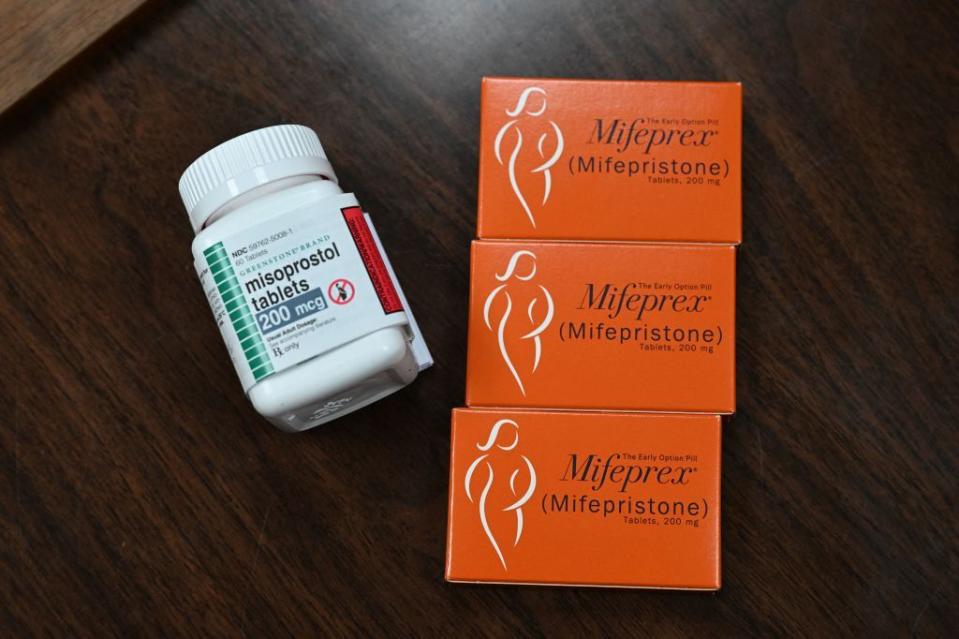
“We know that many providers plan to continue to provide mifepristone prescriptions, while others are also prepared to switch to a misoprostol-only regimen, which we know is also safe and effective,” Plan C’s Wells says. “We also know that mifepristone plus misoprostol pills will remain available through the network of alternate suppliers we list in our Guide to Pills, so we are shoring up that information in anticipation of the ruling.”
Planned Parenthood, one of the largest and most well-known abortion providers in the U.S., for its part has pledged its continued commitment to doing whatever possible to ensure patients can choose the method of abortion that is best for their circumstances, including medication abortion.
Some legal experts also predict that the Texas ruling could have much farther effects, going well beyond just mifepristone. “The potential broader implications are definitely top of mind for Hey Jane,” Freedman admits. “If a court is able to reverse FDA approval on mifepristone, other crucial medications in reproductive health care could also be threatened.” These could include misoprostol, as well as medications outside of abortion, like birth control—despite continued data showing that these are all safe medications that can be safely distributed by mail.

Even in the event that the Texas decision has no impact on the availability of mifepristone or any other drug, the case has already added misinformation and fear to existing confusion around abortion. Right now, only one in three adults knows medication abortion exists, and providers continue to face an uphill battle when trying to advertise their services or spread awareness about abortion options.
“We constantly have to fight advertising partners to even use words like abortion,” says Monica Cepak, the CMO of Wisp, an online provider of reproductive health care. The telehealth company and countless others that provide information on or access to abortion experience ongoing censorship from social media platforms and regularly see their posts and accounts removed or suppressed simply for sharing medically accurate information.
“We all have to cut through a significant amount of white noise, and it only makes it harder for the person sitting at home to know who’s real and who’s not and where to find real, reliable information on abortion,” Grant, of Carafem, says.
This noise will only get louder, she predicts, if mifepristone’s FDA approval is revoked or further called into question. “Using medication abortion has proven to help people make their own private medical decisions, it expands access to care, reduces stigma, and takes some of the burden off of health care clinics that are already overflowing with demand,” Grant explains. “Taking it away will create a further burden for those trying to receive care and will ultimately have a negative impact on maternal health. So, this is very concerning for those of us who want to provide options to keep people safe and healthy, and it’s not going to stop anytime soon.”
You Might Also Like

