UK approves BioNTech and Pfizer’s COVID-19 vaccine for use 'from next week'
WATCH: UK first in the world to approve BioNTech/Pfizer vaccine
The UK has become the first western country to grant emergency authorisation of the COVID-19 vaccine created by Germany’s BioNTech (BNTX) in partnership with US pharma giant Pfizer (PFE).
The Medicines and Healthcare Regulatory Agency decision is “based on a rolling submission, including data from the Phase 3 clinical study, which demonstrated a vaccine efficacy rate of 95%,” the companies said in a press announcement.
“The government has today accepted the recommendation from the independent Medicines and Healthcare products Regulatory Agency to approve Pfizer-BioNTech’s COVID-19 vaccine for use,” the government said. “The vaccine will be made available across the UK from next week.”
The Pfizer/BioNTech vaccine requires super-cold storage of -70C. However, it can be stored for up to five days at 2-8°C, which is possible in a normal fridge.
READ MORE: BioNTech: The German company behind the potential first COVID-19 vaccine
Since the first priority groups for vaccination are care home residents, who might not be able to travel to a hospital or vaccination centre, being able to store the vaccine at normal fridge temperatures for a few days means it could potentially be brought to them.
The British government will purchase 40 million doses of the vaccine. The first batch of deliveries to the UK will start next week. Health minister Matt Hancock has said that the first 10 million doses will be delivered this year.
“From early next week we will start the programme of vaccinating people against COVID-19 here in this country,” Hancock told Sky News on Wednesday, calling the approval “fantastic news.”
WATCH: Is Pfizer-BioNTech vaccine safe and will it work? - Your questions answered
“Today’s emergency use authorisation in the UK marks a historic moment in the fight against COVID-19. This authorisation is a goal we have been working toward since we first declared that science will win, and we applaud the MHRA for their ability to conduct a careful assessment and take timely action to help protect the people of the UK,” said Albert Bourla, the chairman and chief executive officer of Pfizer.
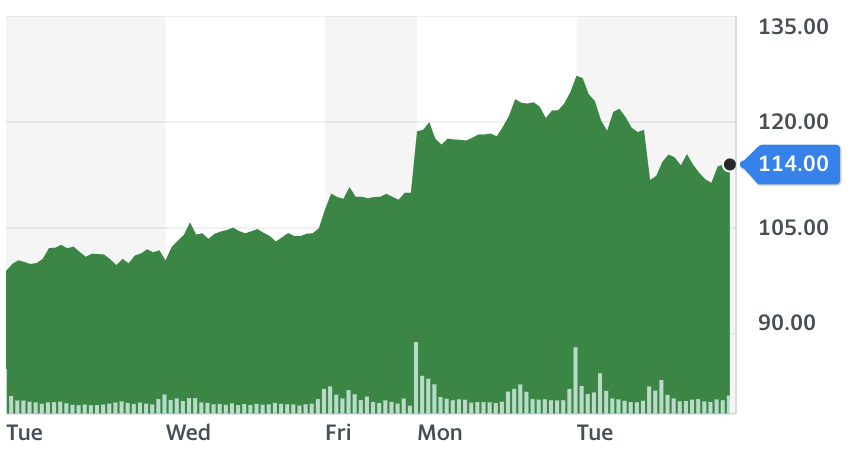
“The emergency use authorisation in the UK will mark the first time citizens outside of the trials will have the opportunity to be immunised against COVID-19,” said BioNTech founder and CEO Ugur Sahin.
“The data submitted to regulatory agencies around the world are the result of a scientifically rigorous and highly ethical research and development programme,” Sahin said.
BioNTech and Pfizer also submitted their vaccine for conditional approval with the European Medicines Agency (EMA) on Tuesday (1 December). They also applied for emergency approval to the US Food and Drugs Administration on 20 November.
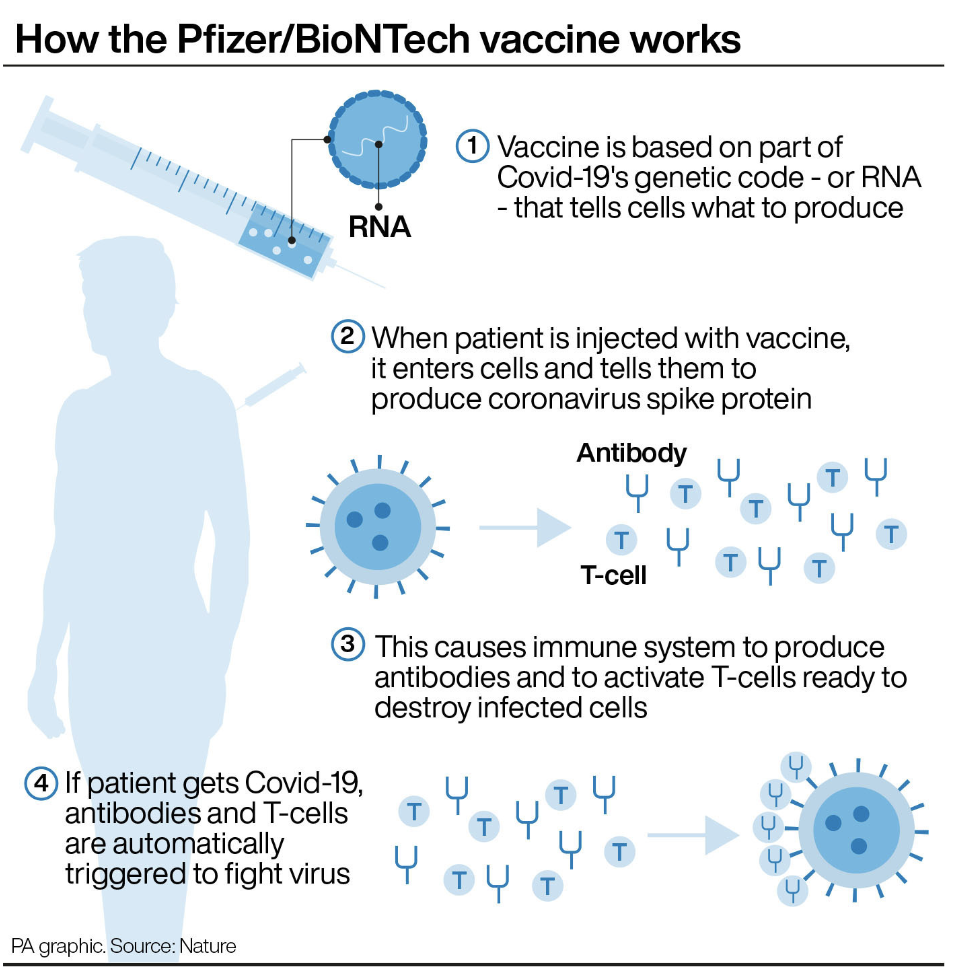
READ MORE: Pfizer says their COVID-19 vaccine ‘95% effective in over 65s’
Pfizer/BioNTech announced on 18 November that their vaccine protected 95% of people from COVID-19, and that it was equally effective across all ethnicities.
The EMA said on Monday that it will decide on authorisation of the Pfizer/BioNTech vaccine by 29 December. Then the European Commission will issue final authorisation for the vaccine to be distributed to EU countries.
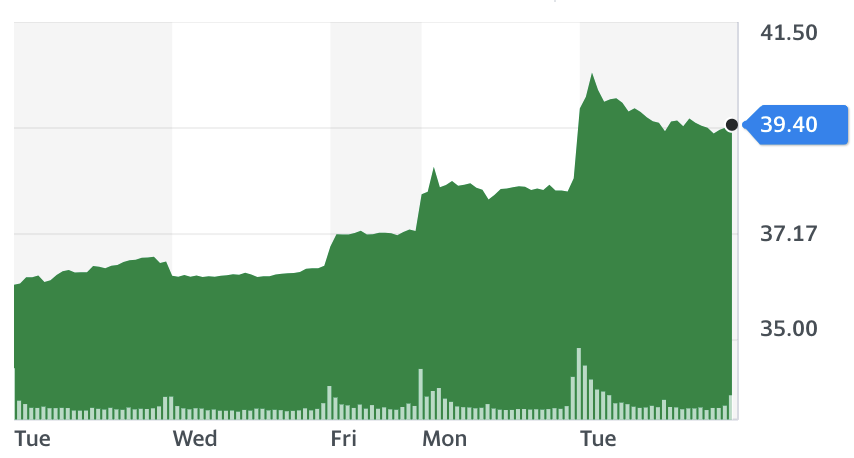
On Monday (30 November), US-based Moderna (MRNA) also submitted its vaccine to the EU and US regulators for emergency approval. Final data analysis from Moderna’s large-scale trial found its vaccine to be 94.1% effective at protecting people from the virus.
The UK government has also bought 7 million doses of the Moderna vaccine.
WATCH: EU plans vaccines as regulator sets approval deadline

