Allarity Therapeutics (NASDAQ: ALLR): Diamond in the Rough

NY City, New York, United States, Aug. 11, 2022 (GLOBE NEWSWIRE) -- Diamond in the Rough
Click here for full length version of article.
Allarity Therapeutics, Inc. (NASDAQ: ALLR) is a clinical stage oncology therapeutics company founded in Denmark in 2004 by Chief Scientific Officer Steen Knudsen, Ph.D. It is pursuing a personalized medicine approach to build upon the success of its Drug Response Predictor (DRP®) companion diagnostic platform to advance oncology therapies, oriented towards combination therapies. The company offers a portfolio of three priority drug candidates, and plans to pursue a Phase Ib/II study in metastatic ovarian cancer starting in 4Q:22 with both stenoparib and dovitinib.

With executive leadership from CEO James G. Cullem, Allarity is taking a new approach to oncology drug development that relies on identification of cancer patients likely to respond to a drug based on an individual’s tumor cell transcriptome. This approach addresses some of the key drawbacks of conventional biomarker approaches, which fail to recognize the multifactorial drivers behind cancer and lack clinical validation. Allarity Therapeutics offers pipeline programs addressing significant markets in metastatic ovarian cancer (mOC), metastatic breast cancer (mBC) and, potentially, metastatic renal cell carcinoma (mRCC), among other indications. We will introduce you to Allarity’s DRP® companion diagnostic platform, its combination therapy approach, and the drivers for the company’s future success.
Drug Response Predictor (DRP®) Platform
The use of biomarkers predicting patient response is widespread. Biomarkers can be based on genetics, mutations, hormone receptor status, presence of enzymes, and tumor markers to name a few. Most of these approaches are one-dimensional, fail to recognize cancer as a mechanistically and biochemically diverse disease and lack clinical validation. To address this shortcoming, Allarity has developed its DRP® platform technology to identify cancer patients that are most likely to benefit from a specific drug.
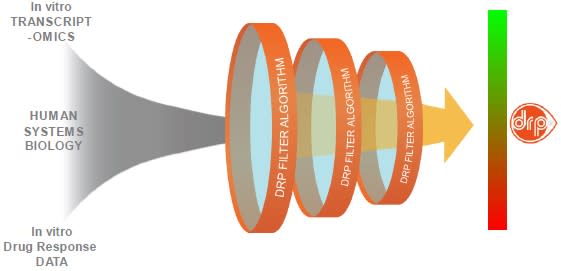
Before a drug-specific DRP® biomarker can be used as a companion diagnostic to select and treat cancer patients, it must be clinically validated using patient tumor biopsies. The process ensures that the biomarker can identify responder patients and improve their therapeutic benefit as compared to patients not selected by the DRP® companion diagnostic.
After passing the arduous validation process, the DRP® companion diagnostic is ready for use in clinical trials with developmental or approved drugs, subject to regulatory requirements and approvals.
Stenoparib and Dovitinib
DRP®’s success has propelled stenoparib and dovitinib into their positions as lead candidates in the company’s pipeline, initially addressing mOC in combination. Combination therapy has emerged as the leading edge of cancer treatment as it addresses the multifactorial nature of a tumor. By employing drugs that use different mechanisms, combination therapy decreases the likelihood that a part of the tumor will escape the action of a drug and proliferate, allowing the cancer to return. Combination therapies also provide synergies that can increase the success of a treatment regimen when used together that exceed the summed benefit when used alone.
Based on the data generated to date and potential mechanism-of-action synergy, Allarity expects to launch a Phase Ib/II combination study in mOC with stenoparib and dovitinib in second line or later patients. The trial should begin in 4Q:22.
Ovarian Cancer
Allarity’s lead indication for its combination approach is second line or later patients in metastatic ovarian cancer. Ovarian cancer ranks 5th in cancer deaths for women and will affect an estimated 20,000 this year in the United States.1 Around the globe, it is ranked 8th for women with more than 313,000 new cases in 2020.2 About 90% of ovarian cancer is in epithelial tissue and most of these cases are high grade and advanced.
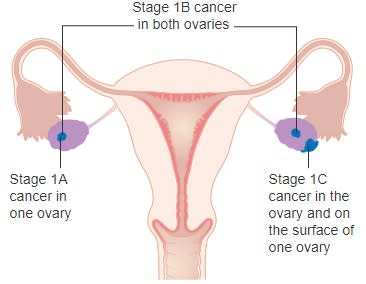
Risk factors for ovarian cancer include smoking, hormone therapy, early menstruation, late menopause, obesity and family history. Symptoms are not obvious, but pelvic pain, back pain, and gastrointestinal-related discomfort are associated with the disease. Treatment for ovarian cancer includes surgery and chemotherapy as well as newer approaches including immunotherapy and targeted therapy. Immune checkpoint inhibitors, such as pembrolizumab (KEYTRUDA®), are therapeutic options. Antiangiogenic agents in the form of monoclonal antibodies and PARP inhibitors are also used to treat the tumor.
A Brilliant Cut
With multiple clinical-stage drug candidates, Allarity Therapeutics avoids being reliant on the success of any single program, and presents investors with several opportunities to drive the company’s future success.
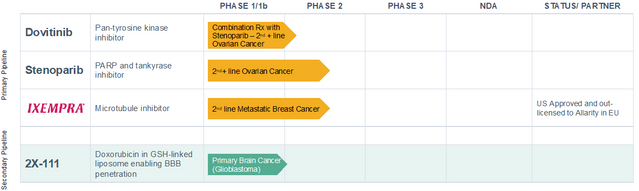
Allarity’s lead therapeutic programs benefit from the synergies of combination therapy and the precision medicine approach achieved through DRP® companion diagnostics, which identify those cancer patients that will respond best to a therapeutic, while bypassing treatment for non-responders. Next steps for the oncology company’s lead programs are the start of a Phase Ib/II study first identifying the optimal dose of the combination of stenoparib and dovitinib in mOC, followed by an initial efficacy study. Second line metastatic ovarian cancer is a difficult and serious disease with a material unmet need that will be aided by Allarity’s program if successful. These Phase II assets may advance into pivotal trials guided by DRP® companion diagnostics, providing additional opportunities in oncology and recognition of this diamond in the rough.
~~~~~~~~~~~~~~~~~~~~~~~~~~
DISCLOSURE: The author of this article has received compensation from an issuer, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing written content. John D. Vandermosten, CFA certifies that the views expressed in the research work accurately reflect his personal views about the subject security and issuers. No part of his compensation was, is, or will be directly or indirectly related to the specific recommendations or views contained in the research article.
Lead Image: Source: Source: US Geological Survey
0 Source: Allarity May 2022 Corporate Presentation.
1 Source: American Cancer Society. Cancer Facts & Figures 2022.
2 World Cancer Research Fund International. Ovarian Cancer Statistics.
3 Source: Wikimedia Commons, Cancer Research UK
4 Source: Allarity May 2022 Corporate Presentation, as modified by Allarity press release on August 2, 2022.
For the original news story, please visit https://www.prdistribution.com/news/allarity-therapeutics-nasdaq-allr-diamond-in-the-rough/9247490
CONTACT: Full Name : John Vandermosten Company : Unboxing Biotech Phone Number : (240) 778-4204 Website : http://www.unboxingbiotech.com Email : contact@unboxingbiotech.com

