The 10 most promising coronavirus treatments that could help curb the pandemic, even without a vaccine

Yuqing Liu/Business Insider
Hundreds of experimental COVID-19 treatments have entered clinical testing in the last few months.
Two — remdesivir and convalescent plasma — have gotten emergency authorization from the FDA.
Here are the 10 candidates the scientific community finds most promising, including antibody therapeutics, plasma, and interferon drugs.
The world has eradicated only one human infection: smallpox.
So the novel coronavirus isn't likely to disappear from the population completely — even if a vaccine is quickly developed, approved, and distributed.
Under the most optimistic scenario, the US could be ready to manufacture a vaccine by the end of 2020. But by then, the virus is expected to have killed a projected 171,500 Americans on top of the current death toll of more than 200,000, according to models from the University of Washington.
Plus, even the most promising vaccine candidates in development face challenges. In September, AstraZeneca paused its phase 3 vaccine trial after a participant appeared to have an adverse reaction. The trial has resumed in the UK, though the US study of the vaccine remains on hold. Even without delays or setbacks, the difficult task of mass-producing and distributing a vaccine mean herd immunity is a long way off.
That makes one thing certain: We need treatments and therapeutics, no matter what else happens.
So far, two have been approved for emergency use by the Food and Drug Administration: remdesivir and convalescent plasma.
Hundreds more have entered clinical testing, from new medications designed to fight the virus directly to repurposed drugs that inhibit a runaway immune response.
"What we really need are drugs that, when given early, can prevent a symptomatic person from requiring hospitalization or very dramatically diminish the time that they're symptomatic," Dr. Anthony Fauci, the director of the National Institute of Allergy and Infectious Diseases, told the Facebook founder Mark Zuckerberg in July.
We've rounded up the 10 treatments that the scientific community thinks are most promising right now. The table below lists them in alphabetical order.
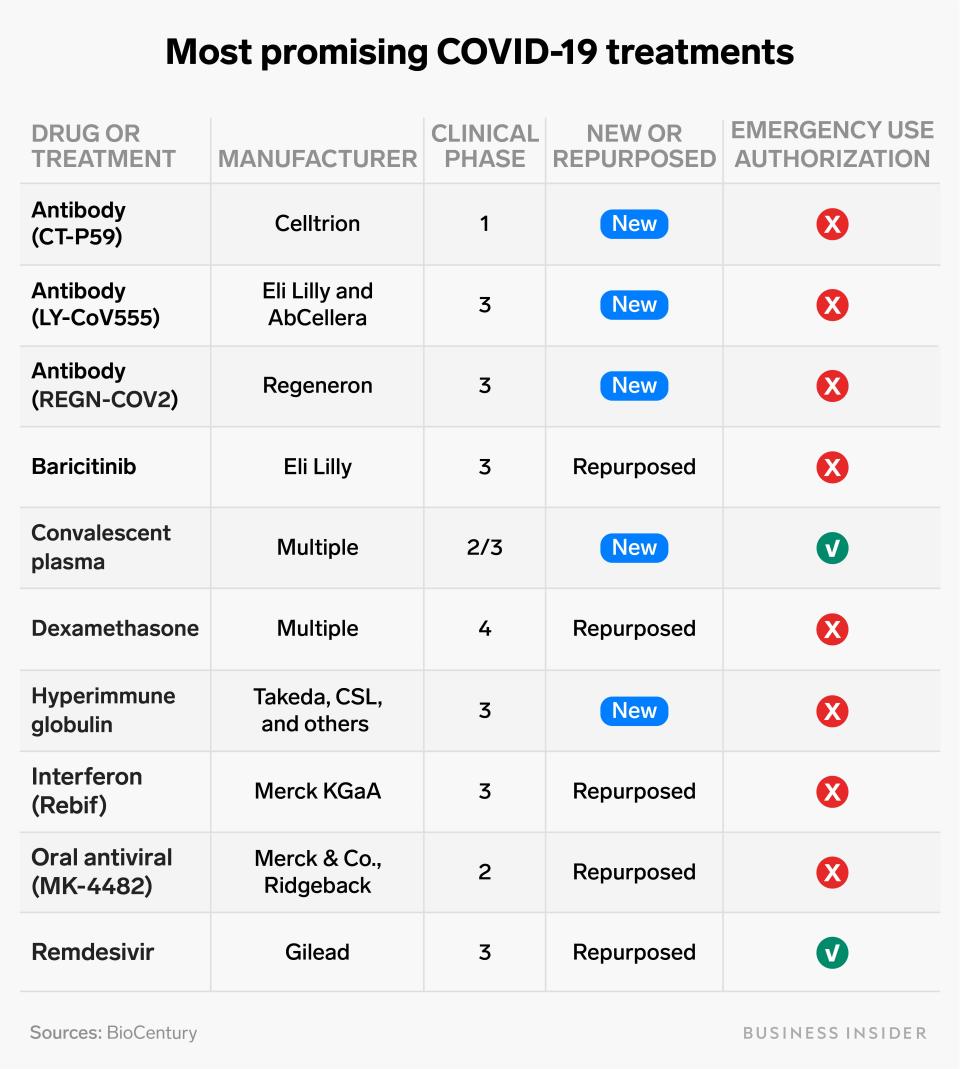
Yuqing Liu/Business Insider
Here's a rundown of the leading candidates.
Celltrion's antibody therapeutic uses the body's natural defenses to fight infection
Many people who get the coronavirus develop neutralizing antibodies — our body's natural response to a foreign pathogen. Fauci has called them "the gold standard of protection against a viral infection."
To make a therapeutic, scientists hunt for the most potent antibodies among a large pool collected from blood donations or animal testing. Then they clone and produce those antibodies at massive scale. There are now about 50 research programs to develop antibody drugs for COVID-19.
Celltrion, a South Korean biotechnology company, launched a human trial for its antibody drug, CT-P59, in mid-July. The study is small — 32 healthy volunteers — and will focus on the drug's safety, but the company expects to know whether it's effective by the end of 2020.
The company also plans to launch studies in Europe by the end of the year to see whether the drug can treat mild and moderate cases. Another trial to test Celltrion's drug as a preventive tool should have results by the first quarter of 2021.
"Celltrion should never be underestimated," Ronny Gal, an industry analyst at AllianceBernstein, wrote to investors in July, adding that the company has a "massive manufacturing footprint" and a demonstrated ability to create antibody drugs.
Eli Lilly is testing its antibody drug in nursing homes
Eli Lilly, a $143 billion pharma giant, is best known for selling insulin. The company has two coronavirus antibody candidates in human testing.
Its most advanced is LY-CoV555, developed with AbCellera, a small Canadian biotech company. In August, Eli Lilly started a late-stage clinical trial to test the drug in residents and staff members of nursing homes. The company said the trial would enroll up to 2,400 people to see whether the medicine can prevent infections in places that have recently had confirmed cases.

Reuters
Data showing whether the drug actually works should be available in the fourth quarter of 2020, Eli Lilly said, adding that it hopes to have several hundred thousand doses of a treatment available by the end of this year.
In an interview with Insider, Bill Gates said antibody drugs could "cut the death rate quite dramatically" among coronavirus patients.
Regeneron is a leader in crafting antibody therapeutics
The biotech company Regeneron has engineered mice to produce antibodies that mimic the human immune system through a technology platform called VelocImmune. That technology has produced several approved drugs, including an Ebola antibody drug that helps to lower patients' risk of death.
In June, Regeneron started clinical trials for REGN-COV2, its two-antibody cocktail. These initial trials focus on patients who already have the coronavirus — there's one for hospitalized patients and one for less severe, non-hospitalized cases. The company said in July that data from the first 30 patients suggested that the drug could be safe for humans.
On September 29, Regeneron released a description of early results from the first 275 volunteers in the non-hospitalized patient study. Chief Scientific Officer George Yancopoulos said the data confirmed the company's belief that the drug is most likely to benefit people who did not mount an immune response before taking the drug.
REGN-COV2 is also being studied as a preventive treatment; Regeneron launched a trial for that purpose in July. The study is designed to test people who have close exposure to the virus, such as someone living with an infected person. Researchers expect to enroll about 2,000 participants across 100 sites.
Eli Lilly is also testing a rheumatoid arthritis drug
Scientists have a few theories as to why coronavirus cases can quickly become severe. One is that an excess of chemical signals can instruct the body to attack its own cells — what's known as "cytokine storm." A class of anti-inflammatory drugs called Janus kinase (JAK) inhibitors help to calm this storm by blocking cytokine production. They're already used to treat rheumatoid arthritis.
One of these arthritis drugs, Eli Lilly's baricitinib, is approved to treat adults with moderate to severe rheumatoid arthritis in more than 70 countries. The drug is now showing early promise as a COVID-19 treatment when combined with another drug: Gilead Sciences' remdesivir.
The drug combo entered a phase-three clinical trial in May that involves more than 1,000 hospitalized COVID-19 patients. In September, Eli Lilly announced that together, baricitinib and remdesivir reduced patients' average recovery time by one day compared to patients who only received remdesivir. The results put the company one step closer to seeking emergency authorization from the FDA.
Convalescent plasma could keep mild cases from getting severe
The FDA issued an emergency authorization for this treatment on August 23.
Convalescent-plasma therapy dates back to the 1918 flu pandemic, when it helped reduce mortality among people with acute infections. Since antibodies develop in plasma, the liquid portion of blood, doctors can transfer this plasma intravenously to a coronavirus patient. That should prompt the new patient to mount an antibody response, which could prevent a mild case from becoming more severe.
So far, a Mayo Clinic study that's still awaiting peer review found that plasma therapy could reduce mortality among hospitalized patients by 57%. And in a study involving 5,000 adults with severe or life-threatening cases, Mayo Clinic researchers determined that the treatment was relatively safe.

Asaad Niazi/AFP/Getty Images
The FDA's emergency authorization allows doctors to administer the treatment earlier in the course of an infection, when it's believed to be most effective.
But Dr. Thomas File, president of the Infectious Diseases Society of America, said the available data on the treatment is still insufficient.
"While the data to date show some positive signals that convalescent plasma can be helpful in treating individuals with COVID-19, especially if given early in the trajectory of disease, we lack the randomized controlled trial data we need to better understand its utility in COVID-19 treatment," File said in a statement on August 23.
More than 2,700 hospitals have already administered plasma therapy through an expanded-access program led by the Mayo Clinic. The program has delivered plasma to more than 105,000 patients, its website says — but it discontinued enrollment once the FDA authorization was announced.
Dexamethasone is a cheap option for patients with acute cases
One of the biggest surprises in COVID-19 research so far has been dexamethasone. Researchers behind a major UK clinical trial found that the cheap, generic steroid significantly reduced the chance that a critically ill patient would die.
The study's authors said dexamethasone reduced the number of deaths by one-third for people on ventilators and by 20% among patients with COVID-19 who were getting extra oxygen.
Reuters
"When you have late disease, it's less the virus doing damage than the aberrant inflammatory response, which is the reason why dexamethasone works," Fauci said in July.
The UK government has authorized dexamethasone to be used as a standard treatment for seriously ill COVID-19 patients. US treatment guidelines also recommend using the steroid in patients who are on ventilators or need oxygen support, though the FDA has not issued an emergency use authorization.
Hyperimmune globulin could buy time until a vaccine hits the market
In May, a coalition of medical institutions, drug companies, nonprofits, and COVID-19 survivors launched The Fight Is In Us, a campaign to get more recovered coronavirus patients to donate blood. Some of that blood will be used for direct transfusions, and the rest will go toward manufacturing hyperimmune globulin, a drug made from convalescent plasma, the group said.
Creating hyperimmune globulin involves pooling plasma from recovered patients and heat-treating it so that any remaining pathogens are destroyed. The result is a vial of medicine with consistent antibody levels that can easily be administered to patients. The drug focuses on the most common antibody found in the blood: immunoglobulin G, or IgG, which usually confers long-term immunity.
Japan's largest pharmaceutical company, Takeda, and a Pennsylvania biotech called CSL Behring are leading a coalition of 10 drug companies involved in the campaign. They're hoping to determine whether hyperimmune globulin improves outcomes for patients with severe coronavirus cases.
"Our goal here isn't to continue to produce hyperimmune globulin ad infinitum," Christopher Morabito, the head of research and development for plasma-based therapies at Takeda, previously told Business Insider. "Our goal here is to have an effective therapy to bridge us to a point where either the pandemic is over because it dies out, or because there's a vaccine available, or until there are many more effective treatments for patients with this disease."
The coalition hopes to secure FDA approval by the end of 2020.
Merck KGaA's interferon drug is being paired with other promising therapies
Interferons are our bodies' first line of defense against the coronavirus. But early research has suggested that the virus might disable these proteins, leading to an even more aggressive immune response.
Drugmakers are now testing whether a boost of interferons could help ward off severe infection.
Doctors already use Rebif, an interferon injection from the German biopharmaceutical company Merck KGaA, to treat patients who relapse from multiple sclerosis. The company hopes the drug will also prevent the coronavirus from replicating in humans and reduce inflammation.
The drug is being tested in conjunction with other therapies like lopinavir (an HIV antiviral) and remdesivir, which was developed as a treatment for Ebola. The National Institute of Allergy and Infectious Diseases launched a phase 3 trial in early August to see whether Rebif and remdesivir could shorten recovery times in coronavirus patients. The study, which hopes to enroll 1,000 patients, could yield preliminary results this fall.
Merck's antiviral pill could be administered right after someone tests positive
Antivirals work by targeting a virus to keep it from replicating inside the body. Some researchers think that since these drugs are already used to treat the flu and HIV, they could represent the future of coronavirus therapy.
"As soon as somebody comes in with a positive test, bingo, you hit 'em with an antiviral and you're done. That's what I see for the future," Fauci said in August. "Quite frankly, there really is no reason why we couldn't do that. There's no reason why that's not possible. Heck, if we did it for HIV, we can do it for coronavirus."
Pharmaceutical company Merck & Co. is working with a biotech company called Ridgeback Biotherapeutics to develop MK-4482, an antiviral administered in pill form. The drug, originally designed as a flu treatment, has been found to prevent multiple coronaviruses from replicating in mice. Other research has found that it could prevent the novel coronavirus from replicating in human cells inside a laboratory — but that doesn't necessarily mean it will work on patients.
The drug's phase-two trial is set to end in September. Merck plans to launch two phase-three trials that month to study the effects of the drug in both hospitalized coronavirus patients and coronavirus outpatients. The goal is to see whether the treatment shortens symptoms or prevents cases from getting worse.
In a July call with investors, Merck's research chief, Roger Perlmutter, said the company could be ready to deliver "millions of doses" before next year.
Remdesivir was the first treatment approved for emergency use by the FDA
The drug was given emergency authorization to treat COVID-19 on May 1.
Remdesivir had already been tested in Ebola patients and found to be safe for humans. Studies have found that the drug, which the company has been working on since 2009, could help hospitalized COVID-19 patients recover faster.

Gilead Sciences Inc/Handout via Reuters
But research published in the Journal of the American Medical Association found that patients put on a 10-day course of remdesivir didn't fare much better than patients who didn't get the drug at all. Both patient groups died at the same rate, and the remdesivir patients experienced more minor to moderate symptoms and side effects (though they reported fewer severe symptoms overall).
Based on these findings, some medical experts suggested that the widespread use of remdesivir may not be worth the cost. After donating its drug supply for two months, Gilead announced in June that it would charge the governments of developed countries $2,340 for a five-day course of remdesivir.
Gilead submitted remdesivir for official FDA approval to treat COVID-19 in August. The company is also testing an inhaled version of the drug in hopes of making it available to non-hospitalized patients.
Though remdesivir and dexamethasone are considered the two biggest advances in coronavirus treatments so far, the drug landscape is constantly changing. The industry publication BioCentury offers a full list of therapies being developed for COVID-19. It's possible that other medications on that list could edge ahead in the coming months.
This article was initially published in August and has been updated with additional developments in the race to make effective coronavirus vaccines and treatments.
Read the original article on Business Insider

