Hillary Clinton Responds To Biden-Trump Debate Line With A Zinger Of Her Own
Former Vice President Joe Biden seemed to deliver the most memorable line of Tuesday night’s debate with President Donald Trump.
But Trump’s 2016 debate rival, former Secretary of State Hillary Clinton, delivered a zinger of her own in response.
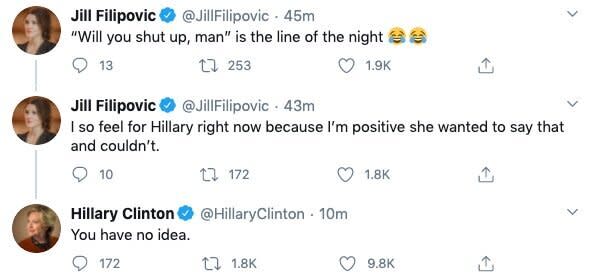
Trump on Tuesday ― as he did with Clinton in 2016 ― repeatedly interrupted during the debate, leading Biden to finally interject: “Will you shut up, man?”
That led to this exchange on Twitter:
Love HuffPost? Become a founding member of HuffPost Plus today.
This article originally appeared on HuffPost and has been updated.

