Fauci Says the FDA Could Approve the COVID Vaccine for Kids Under 5 Next Month
- Oops!Something went wrong.Please try again later.
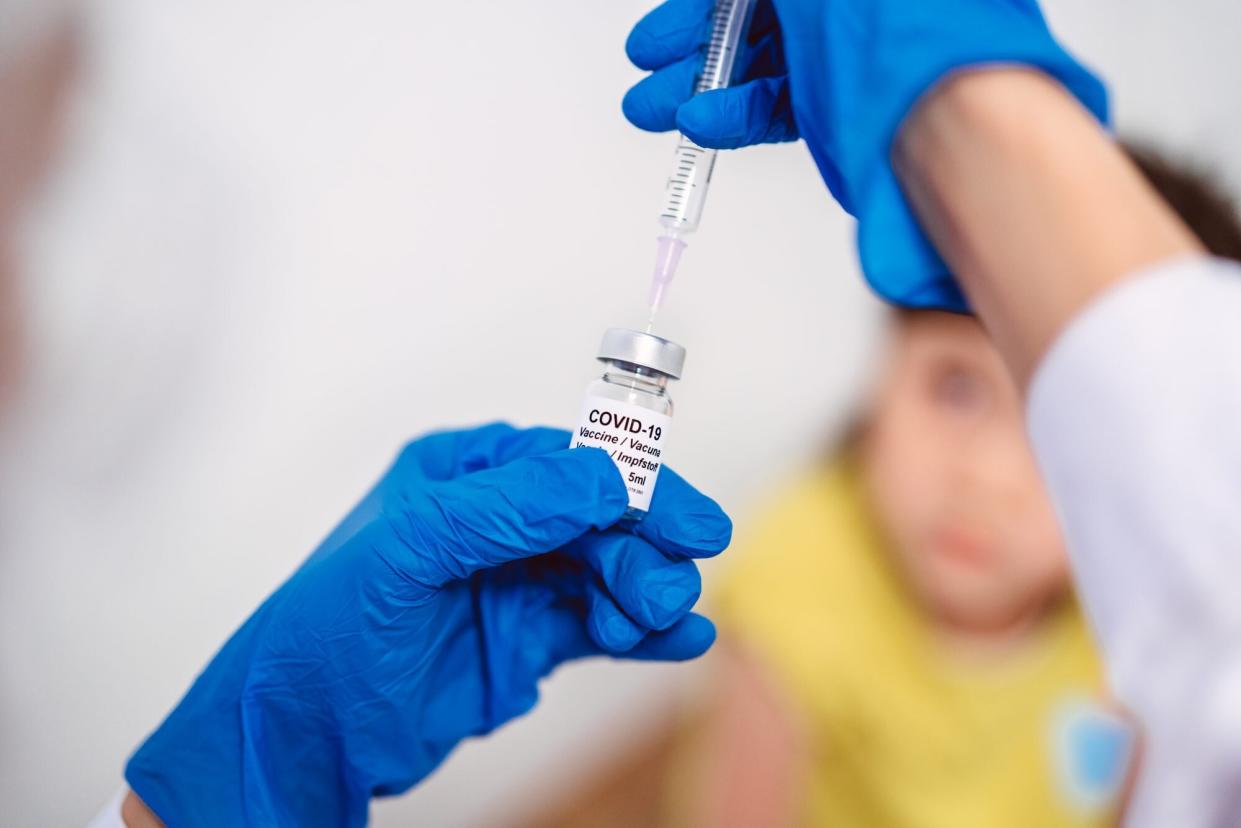
Getty A child getting the COVID-19 vaccine
Children under 5 could be eligible for Pfizer-BioNTech's COVID-19 vaccine within the next month, Dr. Anthony Fauci said Wednesday.
Fauci, the nation's top infectious disease expert, said that it's his "hope" that the Food and Drug Administration approves the vaccine for use in this youngest age group in February.
"My hope is that it's going to be within the next month or so and not much later than that, but I can't guarantee that," he said in an interview with Blue Star Families.
Fauci said that kids under 5 will likely need three doses of Pfizer's vaccine right off the bat, unlike the 2-dose series used with those aged 5 and up. During the first round of clinical trials, two doses alone did not elicit enough of an immune response in 2- to 4-year-olds and Pfizer decided to start a new round of trials with a 3-dose series, the company announced in mid-December.
RELATED: Pfizer to Study Third COVID Vaccine Dose in Kids Under 5 After Two Doses Prove Insufficient
"We have decided to modify each of the pediatric studies to incorporate a third dose to the series and seek licensure for a three-dose series rather than a two-dose series as originally anticipated," Kathrin Jansen, head of vaccine research at Pfizer, said at the time.
There were no safety concerns, however, and Pfizer said that the 4,500 kids studied for the trials tolerated the vaccine well. Pfizer said they plan to submit their clinical trial data to the FDA and Centers for Disease Control to get the vaccine approved for this age group in early 2022.
RELATED VIDEO: FDA Grants Full Approval to Pfizer's COVID Vaccine
Children under 5 are the only age group that are not yet approved to get a COVID-19 vaccine in the U.S., after the FDA approved Pfizer's vaccine for those 5 to 11 years old in November. Many parents are anxious to see the vaccine approved for this youngest age group and protect them against COVID-19.
RELATED: What Parents Should Know as Pediatric COVID Hospitalizations Soar, According to an ER Physician
And with the highly-contagious omicron variant causing soaring numbers of cases in the U.S., pediatric COVID-19 cases have hit record levels. On Jan. 7, CDC Director Dr. Rochelle Walensky said that since mid-December, when omicron started to spread, the hospitalization rate for kids under 5 jumped to more than 4 in 100,000 children, up from 2.5 in 100,000.
"Pediatric hospitalizations are at their highest rate compared to any prior point in the pandemic," Walensky said.
The hospitalization rate for children aged 5 to 17 was around 1 per 100,000 kids, Walensky said, and kids who are unvaccinated are far more likely to be in the hospital than those who have received the vaccine.
As of Jan. 20, 54.7% of kids aged 12 to 18 are fully vaccinated, and only 19% of those 5 to 11, according to the CDC.
As information about the coronavirus pandemic rapidly changes, PEOPLE is committed to providing the most recent data in our coverage. Some of the information in this story may have changed after publication. For the latest on COVID-19, readers are encouraged to use online resources from the CDC, WHO and local public health departments. PEOPLE has partnered with GoFundMe to raise money for the COVID-19 Relief Fund, a GoFundMe.org fundraiser to support everything from frontline responders to families in need, as well as organizations helping communities. For more information or to donate, click here.

